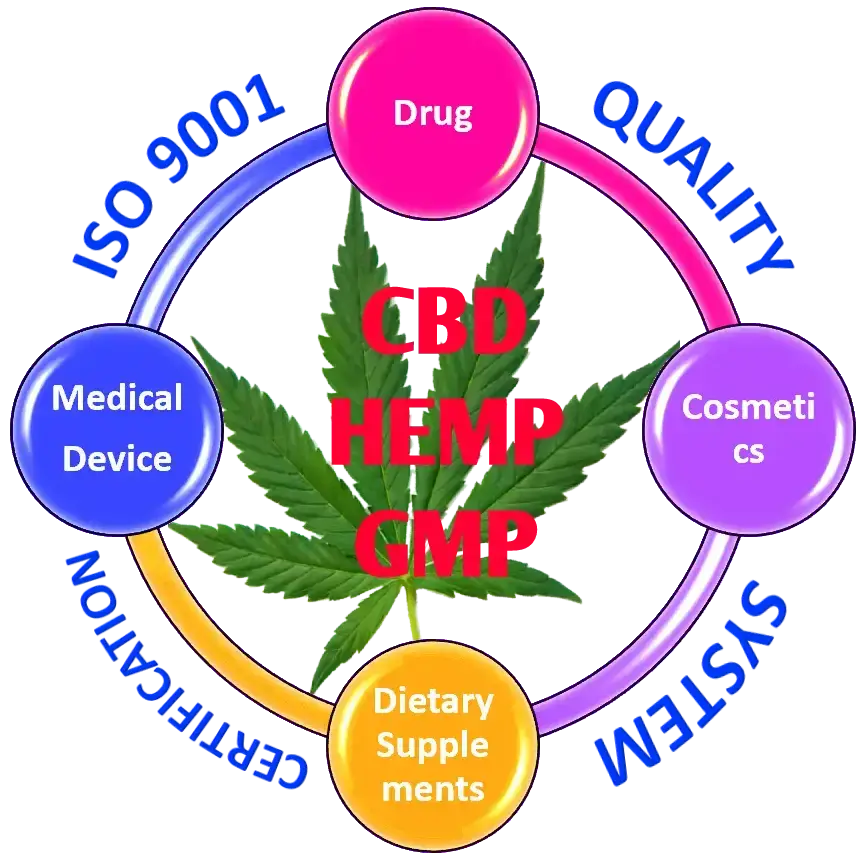
ISO CERTIFICATION - CBD - HEMP - CANNABIS
CBD products are not federally regulated altogether; instead, it is regulated by different agencies like FDA and TTB depends on their intended use and labeling. If your product is a CBD supplement it is regulated by US FDA and you need to comply with supplement GMP regulations, In case your product is CBD cream for pain relief it is regulated by FDA as drug and comes under Drug GMP requirements. If you are manufacturing CBD Vapor for use in tobacco products, it is regulated by TTB.
Which Certification is best for CBD products? ISO or GMP?
If you have multiple products under different category like drug, supplement, cosmetics, vapor etc. or plan to expand existing product line with various products, it is better to implement a quality system which cover all product categories. In such situation ISO 9001 certification is the best option, which is more customizable and flexible.
If you have only one product line with same category of products, it is better to comply with that product category GMP (e.g., Drug gmp, supplement gmp).
Benefits of ISO Certification for CBD and Hemp.
CBD or hemp products regulations are still shaded, and there is no clear-cut guidance for which quality system need to adopt. There is a good chance to regulate all products in Cannabis industry under one umbrella with a new set of GMP regulations. The benefits of ISO 9001 Certification includes.
- ISO 9001 standard is Customizable.
- A transition to new GMP is easy.
- One-size-fits-all certification.
- Worldwide accepted certification.
- Exhibit product quality.
- Boost company credibility.
LMG offers ISO 9001 Certification assistance to Chicago and suburbs from Aurora, Illinois and New York & surrounding states from New York. we provide complete assistance from ISO implementation through certificate issuance.
Our Consulting Services includes
For more information, Please contact us with detailed information.
LIBERTY MANAGEMENT GROUP LTD.
Chicago
75 Executive Drive, Suite 114
Aurora, IL - 60504
Phone : (630) 270-2921
Fax : (815) 986-2632
E-mail : info@libertymanagement.us
New York
100 Duffy Avenue
Hicksville, NY 11801
Phone : (516) 244-2376
E-mail : newyork@libertymanagement.us