GMP for CBD - Hemp – Cannabis Industry
GMP stands for Good Manufacturing Practice, it is a quality system for different industries regulated by governmental and other agencies. The GMP regulations contains the minimum requirements for the manufacturing, processing, and packaging of regulated products. In the USA, GMP is developed by US FDA and is described in title 21 of code of federal regulations. The industries regulated by US FDA are Food - including food additives, Drug - Human and Animal, Biologics, Cosmetics and Medical Device.
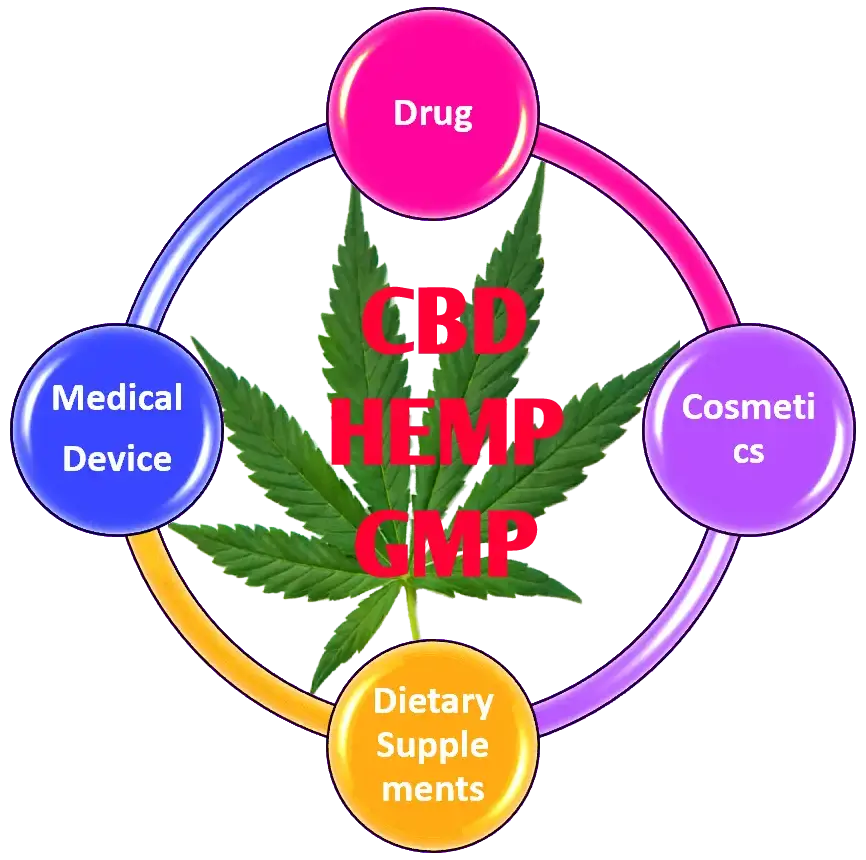
What is GMP for CBD or Hemp?
The GMP for CBD or Hemp is still in the gray area, but if hemp or CBD is used as an ingredient in FDA regulated product or it is marketed as an FDA regulated product; then it comes under FDA regulations and need to comply with corresponding GMP regulations. below examples will explain which GMP is applicable for your product.
if CBD is used as an active ingredient in a pain-relieving cream, it is regulated as a drug and you need to comply with Drug GMP. At present FDA did not include CBD in their OTC Monograph list, so you need prior FDA approval to market it as a drug.
Suppose you are marketing CBD or hemp as a dietary supplement, it comes under food / dietary supplement regulations and you need to comply with food / supplement GMP which is different from Drug GMP. As per latest FDA guidance, FDA is not approving CBD or Hemp products as a dietary supplement.
Same as, if you are using CBD in a cosmetic, it is also regulated by FDA and you need to comply with cosmetic GMP regulations. FDA may inspect the cosmetic facility to make sure the GMP requirements or FDA can review the product labeling to make sure the compliance.
LMG offers GMP and ISO 9001 Certification assistance to Chicago and suburbs from Aurora, Illinois and New York & surrounding states from New York. Our experianced consultants can assist you with.
- Quality Manual
- Quality System procedures
- QMS Implementation
- Internal Audit
- Certification body selection
- Certification body audit
Our Consulting Services includes
For more information, Please contact us with detailed information.
LIBERTY MANAGEMENT GROUP LTD.
Chicago
75 Executive Drive, Suite 114
Aurora, IL - 60504
Phone : (630) 270-2921
Fax : (815) 986-2632
E-mail : info@libertymanagement.us
New York
100 Duffy Avenue
Hicksville, NY 11801
Phone : (516) 244-2376
E-mail : newyork@libertymanagement.us