ISO 9001 Certification Process
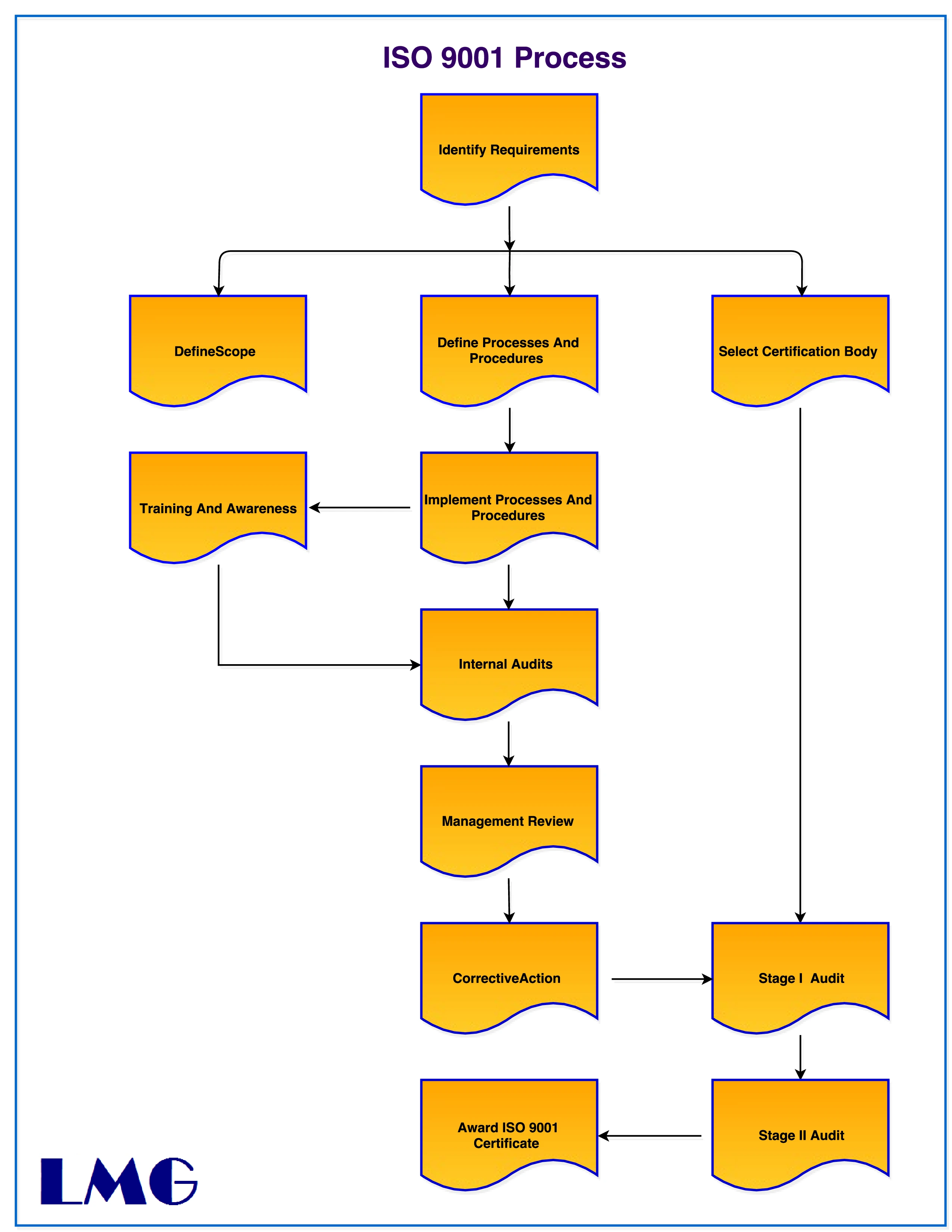
The ISO 9001 certification process is the procedure by which a company or organization is assessed to determine if it meets the requirements of the ISO 9001 quality management standard. The ISO certification process typically involves the following steps:
- Determine if ISO 9001 certification is necessary: Organizations may pursue ISO 9001 certification for a variety of reasons, including regulatory requirements, customer demands, or to improve their own internal operations. The first step is to determine if certification is necessary and if it aligns with the organization's goals.
- Familiarize yourself with the ISO 9001 standard: This involves understanding the requirements of the standard, as well as the certification process.
- Gap analysis: This is an assessment of your organization's current quality management system against the requirements of the ISO 9001 standard to identify any gaps that need to be addressed.
- Implement ISO 9001 quality management system(QMS): The ISO 9001 standard outlines the requirements for a QMS, which is a set of processes and procedures designed to ensure consistent quality in an organization's products or services. Organizations must develop and implement a QMS that meets the requirements of the ISO 9001 standard.
- Conduct an internal audit: Before seeking certification, an organization must conduct an internal audit of its QMS to ensure that it meets the requirements of the ISO 9001 standard. This audit should be conducted by trained personnel who are not directly involved in the QMS.
- Select a certification body: An organization must select a certification body, also known as a registrar or certification body, to conduct the certification audit.
- Schedule the certification audit: The certification audit is typically conducted in two stages.
- Stage 1 Audit, also known as a document review, the certification body will review the organization's QMS documentation to ensure that it meets the requirements of the ISO 9001 standard.
- Stage 2 Audit, also known as certification audit, the certification body will conduct an on-site audit to verify that the QMS is being implemented effectively.
- Receive the certification: If the organization passes the certification audit, the certification body will issue a certificate of conformity, indicating that the organization's QMS meets the requirements of the ISO 9001 standard. The certificate is valid for three years, after which the organization must undergo a recertification audit.
It's worth noting that ISO 9001 certification is not mandatory, but it can provide organizations with several benefits, including improved efficiency, increased customer satisfaction, and a competitive advantage in the marketplace. The ISO 9001 certification process can be complex, and it's recommended to work with an experienced consultant to ensure successful certification.
LMG offers ISO 9001 Certification assistance to Chicago and suburbs from Aurora, Illinois, and New York & surrounding states from New York. We provide complete service from ISO 9001 implementation through certificate issuance; our experienced consultants assist you with
- Quality Manual
- Quality System procedures
- QMS Implementation
- Internal Audit
- Certification body selection
- Certification body audit
Our Consulting Services include
For more information, Please contact us with detailed information.
- ISO 9001 Certification Cost
- ISO 9001 Consultant - Chicago
- How do I get ISO 9001 Certificate?
- ISO 9001 Certification vs Implementation
- ISO 9001 Certification or Approval?
- Transport industry ISO 9001 Certification
- ISO 13485 Certification
- ISO Certification - FAQ
- Hand Sanitizer GMP
- GMP Certification
- 21 CFR 820
- CE Marking
- US FDA
LIBERTY MANAGEMENT GROUP LTD.
Chicago
75 Executive Drive, Suite 114
Aurora, IL - 60504
Phone : (630) 270-2921
Fax : (815) 986-2632
E-mail : info@libertymanagement.us
New York
100 Duffy Avenue
Hicksville, NY 11801
Phone : (516) 244-2376
E-mail : newyork@libertymanagement.us